Punjab State Board PSEB 11th Class Chemistry Book Solutions Chapter 4 Chemical Bonding and Molecular Structure Textbook Exercise Questions and Answers.
PSEB Solutions for Class 11 Chemistry Chapter 4 Chemical Bonding and Molecular Structure
PSEB 11th Class Chemistry Guide Chemical Bonding and Molecular Structure InText Questions and Answers
Question 1.
Explain the formation of a chemical bond.
Answer:
A chemical bond is defined as an attractive force that holds the constituents (atoms, ions etc.) together in a chemical species.
Various theories have been suggested for the formation of chemical bonds such as the electronic theory, valence shell electron pair repulsion theory, valence bond theory, and molecular orbital theory.
A chemical bond formation is attributed to the tendency of a system to attain stability. It was observed that the inertness of noble gases was because of their fully filled outermost orbitals. Hence, it was postulated that the elements having incomplete outermost shells are unstable (reactive). Atoms, therefore, combine with each other and complete their respective octets or duplets to attain the stable configuration of the nearest noble gases. This combination can occur either by sharing of electrons or by transferring one or more electrons from one atom to another. The chemical bond formed as a result of sharing of electrons between atoms is called a covalent bond. An ionic bond is formed as a result of the transference of electrons from one atom to another.
![]()
Question 2.
Write Lewis dot symbols for atoms of the following elements: Mg, Na, B, O, N, Br.
Answer:
Mg : There are two valence electrons in Mg atom (2, 8, 2). Hence, the Lewis dot symbol of Mg is :
![]()
Na : There is only one valence electron in an atom of sodium (2, 8,1). Hence, the Lewis dot Symbol is :
![]()
B : There are three valence electrons in Boron atom (2, 3). Hence, the Lewis dot symbol is :
![]()
0 : There are six valence electrons in an atom of oxygen (2, 6). Hence, the Lewis dot symbol is :
![]()
N: There are five valence electrons in an atom of nitrogen (2,5). Hence, the Lewis dot symbol is :
![]()
Br : There are seven valence electrons in bromine (2, 8, 18, 7). Hence, the
Lewis dot symbol is :
![]()
Question 3.
Write Lewis symbols for the following atoms and ions: S and S2-; Al and Al3+; H and H–
Answer:
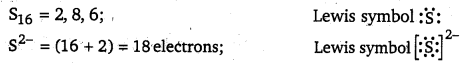

Question 4.
Draw the Lewis structure for the following molecules and ions :
H2S, SiCl4, BeF2, \(\mathrm{CO}_{3}^{2-}\), HCOOH
Answer:
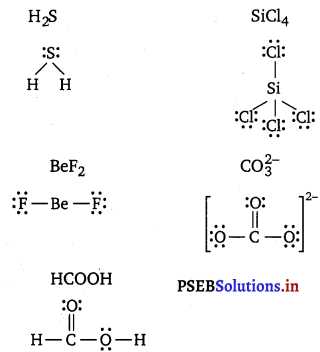
Question 5.
Define octet rule. Write its significance and limitations.
Answer:
Octet rule : Atoms can combine either by transfer of valence electrons from one atom to another (gaining or losing) or by sharing of valence electrons in order to have an octet in their valence shell.
Significance of octet rule : It help to explain why different atom combine with each other to form ionic or covalent compounds. Limitations of the Octet Rule
Although octet rule can explain the formation of a large number of compounds but it has many exceptions also, which are discussed below :
(i) Electron deficient molecules : There are some molecules in which the central atom is surrounded by less than eight electrons, i.e., their octet is incomplete. Elements having less than four valence electrons generally form molecules of this category.
e.g., BeCl2, BF3, AlCl3, LiCl, BeH2 etc.
(ii) Odd electron molecules : Molecules like NO, NO2, O2 etc., are examples of such molecules in which bonded atoms have odd number of electron (usually 3) in between them. That’s why these are called odd electron molecules.
In case of these molecules, the octet rule is not satisfied for all the atoms, e.g.,

Species with one unpaired electron are called free radicals. These are paramagnetic and most of them are generally unstable.
(iii) Electron rich molecules : Elements of the third and higher periods of the periodic table, because of the availability of d orbitals can expand their covalency and -can accommodate more than eight valence electrons around the central atom. This is referred as expanded octet. Here, also the octet rule is not applicable, e.g., PF5 (10 electrons around P atom), SF6 (12 electrons around S atom), H2SO4 (12 electron around S atom).
Compounds having expanded octet are also termed as hypervalent compounds.
(iv) Other drawbacks : Other drawbacks of this theory are as follows:
1. Octet rule is based on the inertness of noble gases but some noble gases like xenon and krypton form several compounds with oxygen and fluorine like. XeF2, XeF4, XeF6, XeOF4, XeO2F2, KrF2 etc.
2. It does not tell anything about the shapes of molecules and their relative stabilities.
3. It fails to explain the paramagnetic behaviour of oxygen. (Which should be diamagnetic according to this rule but it is infact paramagnetic in nature).
![]()
Question 6.
Write the favourable factors for the formation of ionic bond.
Answer:
The favourable factors for ionic bond formation are as follows :
(i) Low ionization enthalpy of element forming cation.
(ii) More negative electron gain enthalpy of element forming anion.
(iii) High lattice energy of the compound formed.
Question 7.
Discuss the shape of the following molecules using the VSEPR model:
BeCl2, BCl3, SiCl4, ASF5, H2S, PH3
Answer:
According to VSEPR theory, the shape of a molecule depends upon the number of valence shell electron pairs (bonded or non-bonded) around the central atom. Pairs of electrons in the valence shell repel each other. The order of their repulsion is as follows :
Ip -Ip > Ip -bp > bp – bp
(i) BeCl2 or
![]()
The central atom Be has only 2 valence electrons which are bonded to Cl, so there are only 2 bond pairs and no lone pairs. It is of the type AB2 and hence, the shape is linear.
(ii) BCl3:
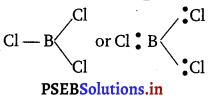
The central atom B has only 3 valence electrons which are bonded with three Cl atoms, so it contains only 3 bond pairs and no lone pair. It is of the type AB3 and hence, the shape is trigonal planar.
(iii) SiCl4 :

Similarly, the central atom Si has only 4 bond pairs and no lone pair. It is of the type AB4 and hence, the shape is tetrahedral.
(iv) AsF5:
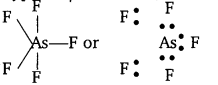
The central atom As has only 5 bond pairs and no lone pair. It is of the type AB5 and hence, the shape is trigonal bipyramidal.
(v) H2S:
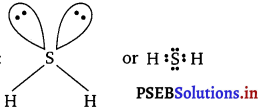
The central atom S has 6 valence electrons. Out of these only two are used in bond formation with two H-atoms while four (two pairs) remains as non-bonding electrons (i.e., lone pairs). So, it contains 2 bond pairs and 2 lone pairs. It is of the type AB2E2 and hence, the shape
is bent or V-shaped.
(vi) PH3
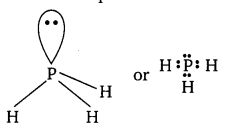
The central atom P has 5 valence electrons. Out of which three are utilised in bonding with H atoms and one pair remains as lone pair. So, it contains 3 bond pairs and one lone pair. It is of the type AB3E and hence the shape is pyramidal.
Question 8.
Although geometries of NH3 and H2O molecules are distorted
tetrahedral, bond angle in water is less than that of ammonia.
Discuss.
Answer:
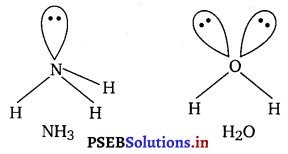
In H2O molecule there, is lone pair-lone pair repulsion due to the presence of two lone pairs of electrons while in NH3 molecule there are only lone pair-bond pair repulsion. According to VSEPR theory the former one is more stronger and hence the bond angle in water is less than that of ammonia.
Question 9.
How do you express the bond strength in terms of bond order?
Answer:
Bond strength represents the extent of bonding between two atoms forming a molecule. The larger the bond energy, the stronger is the bond and the greater is the bond order.
Question 10.
Define the bond length.
Answer:
Bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule.
Bond lengths are expressed in terms of Angstrom (10-10 m) or picometer (10-12 m) and are measured by spectroscopic X-ray diffractions and |
electron-diffraction techniques.
Question 11.
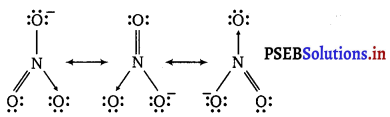
Explain the important aspects of resonance with reference to \(\mathrm{CO}_{3}^{2-}\) the ion.
Answer:
According to experimental findings, all carbon to oxygen bonds in \(\mathrm{CO}_{3}^{2-}\) are equivalent. Hence, it is inadequate to represent \(\mathrm{CO}_{3}^{2-}\) ion by a single Lewis structure having two single bonds and one double bond.
The \(\mathrm{CO}_{3}^{2-}\) ion is best described as a resonance hybrid of the canonical forms I, II and III.
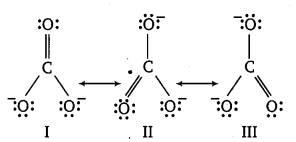
All canonical forms have similar energy, same positions of atoms and same number of bonded and non-bonded pairs of electrons.
![]()
Question 12.
H3PO3 can be represented by structures 1 and 2 shown below. Can these two structures be taken as the canonical forms of the resonance hybrid representing H3PO3? If not, give reasons for the same.

Answer:
The given structures cannot be taken as the canonical forms of the resonance hybrid of H3PO3, because the positions of the atoms have been changed.
Question 13.
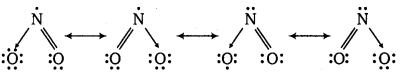
Write the resonance structures for SO3, NO2 and \(\mathrm{NO}_{3}^{-}\)
Answer:
The resonance structures are :
(a) SO3:
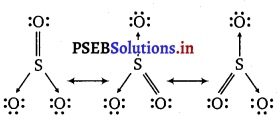
(b) NO2:
(c) \(\mathrm{NO}_{3}^{-}\)
Question 14.
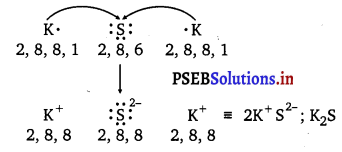
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions: (a) K and S (b) Ca and O (c) Al and N.
Answer:
(a) K and S :
The electronic configurations of K and S are as follows :
K : 2, 8, 8, 1
S : 2, 8, 6
![]()
Sulphur (S) requires 2 more electrons to complete its octet. Potassium (K) requires one electron more than the nearest noble gas i.e., Argon. Hence, the electron transfer can be shown as:
(b) Ca and O :
The electronic configurations of Ca and O are as follows :
Ca : 2, 8, 8, 2
O : 2, 6
Oxygen requires two electrons more to complete its octet, whereas calcium has two electrons more than the nearest noble gas i.e., Argon. Hence, the electron transfer takes place as :
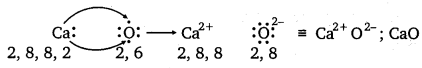
![]()
(c) Al and N :
The electronic configurations of Al and N are as follows :
A1: 2, 8, 3
N : 2, 5
Nitrogen is three electrons short of the nearest noble gas (Neon), whereas aluminium has three electrons more than neon. Hence, the electron transference can be shown as :
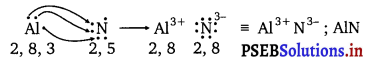
Question 15.
Although both CO2 and H2O are triatomic molecules, the shape of H2O molecule is bent while that of CO2 is linear. Explain this on the basis of dipole moment.
Answer:
According to experimental results, the dipole moment of carbon dioxide is zero. This is possible only if the molecule is linear so that the dipole moments of C—O bonds are equal and opposite to nullify each other.
![]()
Resultant µ = 0 D
On the other hand, H2O molecule is found to have a net dipole moment value of 1.84 D (thoughit is a triatomic molecule as CO2). The value of the dipole moment suggests that the structure of H20 molecule is bent where the dipole moment of O—H bonds are unequal.
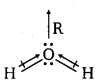
Question 16.
Write the significance/applications of dipole moment.
Answer:
The applications of dipole moment are as follows :
1. In determining the polarity of bonds : As µ = e x d, obviously greater is the magnitude of dipole moment, higher will be the polarity of the bond. This is applicable to molecules containing only one polar bond like HC1, HBr etc. In non-polar molecules like, H2, O2, N2 the dipole moment is zero. It is because there is no charge separations in these molecules [e = 0]. Thus, dipole moment can also be used to distinguish between polar and non-polar molecules.
2. In the calculation of percentage ionic character :
Take the example of HCl. Its µ = 1.03 D
If HCl is 100% ionic, each end would carry charge of one unit
i.e., 4.8 x 10-10 e.s.u.
d (bond length) in H—Cl = 1.275Å
∴ for 100% ionic character, dipole moment will be
µionic = e x d
= 4.8 x 10-10 e.s.u x 1.275 x 10-8 cm
= 6.12 x 10-18 e.s.u cm = 6.12 D
∴ Percentage of ionic character =
![]()
= \(\frac{1.03}{6.12}\) x 100 = 16.83.
3. In determining the symmetry (or shape) of the molecules : Dipole moment is an important property in determining the shape of molecules containing 3 or more atoms. For instant if any molecule possesses two or more polar bonds, it will not be symmetric if it possesses some net molecular dipole moment as in case of water (\(\mu_{\mathrm{H}_{2} \mathrm{O}}\) = 1-84 D) and ammonia (\(\mu_{\mathrm{NH}_{3}}\) = 1.49 D). But if a molecule contains a number of similar atoms linked to a central atom the overall dipole moment of the molecule is found out to be zero, this will imply that the molecule is symmetrical as in the case of CO2, BF3, CH4,CCl4,etc.
Question 17.
Define electronegativity. How does it differ from electron gain enthalpy?
Answer:
Electronegativity is the ability of an atom in a chemical compound to attract a bond pair of electrons towards itself. Electronegativity of any given element is not constant. It varies according to the element to which it is bound. It is not a measurable quantity. It is only a relative number.
On the other hand, electron gain enthalpy is the enthalpy change that takes place when an electron is added to a neutral gaseous atom to form an anion. It can be negative or positive depending upon whether the electron is added or removed. An element has a constant value of the electron gain enthalpy that can be measured experimentally.
Question 18.
Explain polar covalent bond with the help of suitable example.
Answer:
When two dissimilar atoms having different electronegativities combine to form a covalent bond, the bond pair of electrons is not shared equally. The bond pair shifts towards the nucleus of the atom having greater electronegativity. As a result, electron distribution gets distorted and the electron cloud is displaced towards the electronegative atom.
As a result, the electronegative atom becomes slightly negatively charged while the other atom becomes slightly positively charged. Thus, opposite poles are developed in the molecule and this type of a bond is called a polar covalent bond.
For example-In HF, the electron pair is attracted more towards F atom due to its higher electronegativity. HF may be written as
![]()
![]()
Question 19.
Arrange the bonds in order of increasing ionic character in the molecules : LiF, K2O, N2, SO2 and ClF3.
Answer:
More the difference of electronegativity, more the ionic character of the molecules
N2 < SO2 < ClF2 < K2O < LiF.
Question 20.
The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
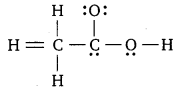
Answer:
The correct Lewis structure for acetic acid is given below :
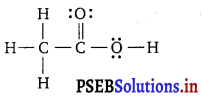
Question 21.
Apart from tetrahedral geometry, another; possible geometry for CH4 is square planar with the four H atoms at the comers of the square and the C atom at its centre. Explain why CH4 is not square planar?
Answer:
Electronic configuration of carbon atom :
6C : 1s2 2s2 2p2
In the excited state, the orbital picture of carbon can be represented as:
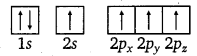
Hence, carbon atom undergoes sp3 hybridization in CH4 molecule and takes a tetrahedral shape.
For a square planar shape, the hybridization of the central atom has to be dsp. However, an atom of carbon does not have d-orbitals to undergo dsp2 hybridization. Hence, the structure of CH4 cannot be square planar. Moreover, with a bond angle of 90° in square planar, the stability of CH4 will be very less because of the repulsion existing between the bond pairs. Hence, VSEPR theory also supports a tetrahedral structure for CH4.
Question 22.
Explain why BeH2 molecule has a zero dipole moment although the Be—H bonds are polar.
Answer:
BeH2 molecule is linear. The two equal bond dipoles point in opposite
directions and cancel the effect of each other.
That is why its dipole moment is zero.

Question 23.
Which out of NH3 and NF3 has higher dipole moment and why?
Answer:
In both molecules i.e., NH3 and NF3, the central atom (N) has a lone pair electron and there are three bond pairs. Hence, both molecules have a pyramidal shape. Since fluorine is more electronegative than hydrogen, it is expected that the net dipole moment of NF3 is greater than NH3. However, the net dipole moment of NH3 (1.46 D) is greater than that of NF3 (0.24 D).
This can be explained on the basis of the directions of the dipole moments of each individual bond in NF3 and NH3. These directions can be shown as :
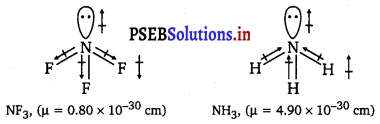
Thus, the resultant moment of the N—H bonds add up to the bond moment of the lone pair (the two being in the same direction), whereas that of the three N—F bonds partly cancels the moment of the lone pair.
Hence, the net dipole moment of NF3 is less than that of NH3.
Question 24.
What is meant by hybridisation of atomic orbitals? Describe the shapes of sp, sp2 , sp3 hybrid orbitals.
Answer:
Hybridisation : It is defined as the mixing of the atomic orbitals belonging to the same atom but having slightly different energies so that a redistribution of energy takes place between them resulting in the formation of new orbitals of equal energies and identical shapes. The new orbitals thus formed are known as Hybrid Orbitals. sp Hybridisation : Here one s and one p orbitals of same atom mix up to
give two sp hybrid orbitals with \(\frac{1}{2}\)s and \(\frac{1}{2}\)p character and linear shape with
bond angle of 180° between them. For example, in BeH2, BeF2 and C2H2, Be and C are sp-hybridised.
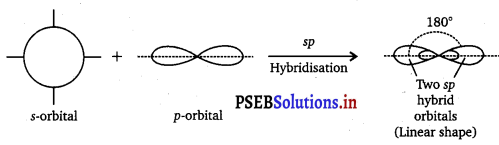
![]()
sp hybridization is also called diagonal hybridization.
sp2 Hybridisation : Here one s and two p-orbitals of same atom mix up to form three sp2hybrid orbitals with \(\frac{1}{3}\)s and \(\frac{2}{3}\)p character. They form Trigonal Planar shapes with an angle of 120° with themselves. For example, in BH3 and BF3, boron is sp2 hybridised and in C2H4, carbon is sp2 hybridised.
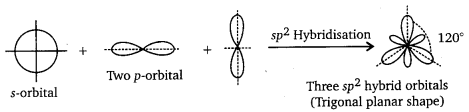
sp3 Hybridisation : Here one s and three p orbitals of same atom mix up to give four sp3 hybrid orbitals with \(\frac{1}{4}\) s character and \(\frac{3}{4}\)p character. They form tetrahedral shapes with angles of 109°, 28′ with themselves. For example, in methane (CH4), ethane (C2H6) and all compounds of carbon containing C—C single bonds, carbon is sp3 hybridised.
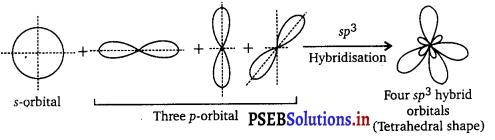
Question 25.
Describe the change in hybridisation (if any) of the A1 atom in the following reaction.
AlCl3 + Cl– → \(\mathbf{A l C l}_{\mathbf{4}}^{-}\)
Answer:
Electronic configuration of A1 in ground state is \(1 s^{2}, 2 s^{2}, 2 p^{6}, 3 s^{2}, 3 p^{\prime}{ }_{x}\) and it is \(1 s^{2}, 2 s^{2}, 2 p^{6}, 3 s^{\prime}, 3 p_{x}^{\prime}, 3 p^{\prime} y\) in excited state.
In the formation of AlCl3 Al undergoes sp2-hybridisation and it is trigonal planar in shape. While in the formation of \(\mathrm{AlCl}_{4}^{-} \), Al undergoes sp3-hybridisation. It means empty 3pz-orbital also involved in hybridisation. Thus, the shape of \(\mathrm{AlCl}_{4}^{-} \) ion is tetrahedral.
Question 26.
Is there any change in the hybridisation of B and N atoms as a result of the following reaction?
BF3 + NH3 → F3B . NH3
Answer:
In BF3, B is sp2 hybridised and in NH3, N is sp3 hybridised. After the reaction hybridisation of B changes to sp3 but that of N remains unchanged.
Question 27.
Draw diagrams showing the formation of a double bond and a triple bond between carbon atoms in C2H4 and C2H2 molecules.
Answer:
Formation of C2H4 (ethylene)
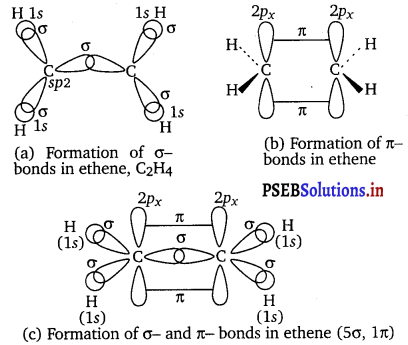
Formation of C2H2 (acetylene)
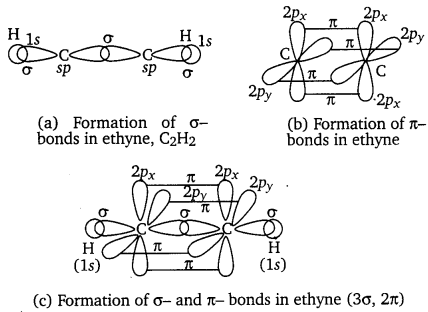
Question 28.
What is the total number of sigma and pi bonds in the following molecules?
(a) C2H2 (b) C2H4
Answer:
(a) The structure of C2H2 can be represented as:
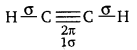
Hence, there are three sigma and two pi-bonds in C2H2.
(b) The structure of C2H4 can be represented as:
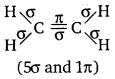
Hence, there are five sigma bonds and one pi-bond in C2H4.
Question 29.
Considering x-axis as the intemuclear axis which out of the following will not form a sigma bond and why?
(a) 1s and 1s (b) 1s and 2px, (c) 2py and 2py (d) Is and 2s.
Answer:
(c) 2py and 2py orbitals will not a form a sigma bond. Taking x-axis as
the intemuclear axis, 2py and 2py orbitals will undergo lateral overlapping, thereby forming a pair bond.
Question 30.
Which hybrid orbitals are used by carbon atoms in the following molecules?
(a) CH3— CH3; (b) CH3—CH = CH2; (c) CH3—CH2—OH;
(d) CH3—CHO; (e) CH3COOH
Answer:
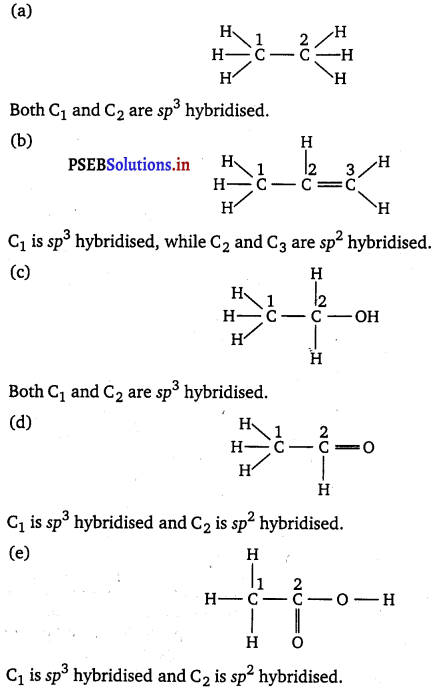
Question 31.
What do you understand by bond pan’s and lone pah’s of electrons? Illustrate by giving one example of each type.
Ans. When two atoms combine by sharing their one or more valence electrons, a covalent bond is formed between them. The shared pairs of electrons present between the bonded atoms are called bond pairs. All valence electrons may not participate in bonding. The electron pairs that do not participate in bonding are called lone pairs of electrons.
For example, in C2H6 (ethane), there are seven bond pairs but no lone pair present.
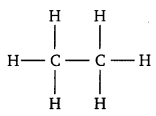
In H2O, there are two bond pairs and two lone pairs on the central atom (oxygen).
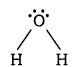
![]()
Question 32.
Distinguish between a sigma and a pi bond.
Answer:
The following are the differences between sigma and pi-bonds :
| Sigma (σ) Bond | Pi (π) Bond | |
| (a) | It is formed by the end to end over lapping (axial over lapping) of atomic orbitals. | It is formed by the lateral overlapping (sideway overlapping) of atomic orbitals. |
| (b) | The orbitals involved in the overlapping are s—s, s—p or p—p. | These bonds are formed by the overlapping of p—p orbitals only. |
| (c) | It is a strong bond. | It is a weak bond. |
| (d) | The electron cloud is symmetrical about the line joining the two nuclei. | The electron cloud is not symmetrical. |
| (e) | It consists of one electron cloud, which is symmetrical about the internuclear axis. | There are two electron clouds lying above and below the plane of the atomic nuclei. |
| (f) | Free rotation about σ bonds is possible. | Rotation is restricted in case of pi-bonds. |
Question 33.
Explain the formation of H2 molecule on the basis of valence bond theory.
Answer:
Consider two hydrogen atoms A and B are approaching each other. Their nuclei are NA and NB and electrons present in them are represented by eA and eB. When the two atoms are far apart, there is no interaction between them but as these approach each other, some new ‘ attractive and repulsive force begin to operate.
Attractive forces generated between
(i) nucleus of one atom and its own electron i.e., NA – eA and NB – eB.
(ii) nucleus of one atom and electron of other atom i.e., NA – eB and NB-eA.
Similarly, repulsive forces originated in between
1. electrons of two atoms i.e., eA – eB
2. nuclei of two atoms NA – NB.
Attractive forces tend to bring the combining atoms close to each other “ whereas repulsive forces tend to push them apart as shown in the figure
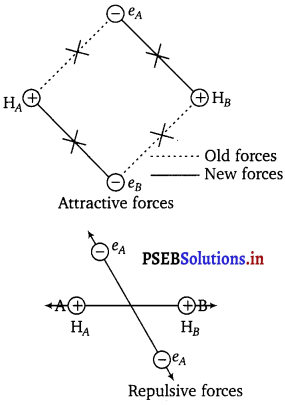
The magnitude of the attractive forces is more than that of the repulsive forces. Hence, the two atoms approach each other. As a result, the potential energy decreases. Finally, a state is reached when the attractive forces balance the repulsive forces and the system acquires minimum energy. This leads to the formation of a dihydrogen molecule.
Question 34.
Write the important conditions required for the linear combination of atomic orbitals to form molecular orbitals.
Answer:
Conditions required for the Combination of Atomic Orbitals
The linear combination of atomic orbitals to form molecular orbitals is possible only when they satisfied the following conditions :
(i) Similar energy of combining atomic orbitals : The combining atomic orbitals must possess the same or nearly the same energy. It means that Is orbital can combine with another Is orbital but not with 2s orbital because the energy of 2s orbitals is appreciable higher than that of Is orbital. However, it is not true in case of very different atoms.
(ii) Similar symmetry of combining atomic orbitals : The combining atomic orbitals must possess the same symmetry about the molecular axis along with the same energy. If the orbitals have same energy but their symmetry is not same, they will not combine e.g., 2pz orbital of one atom can combine with 2pz orbital or 2s orbital of the other atom but not with the 2px or 2py orbitals as their symmetries are different.
(iii) Maximum overlap : The combining atomic orbitals must overlap to the maximum extent. Higher the extent of overlapping, more will be the electron-density between the nuclei of a molecular orbital.
Question 35.
Use molecular orbital theory to explain why the Be2 molecule does not exist.
Answer:
The electronic configuration of Beryllium is 1s2 2s2
The electronic configuration of Be2 molecule (4 + 4 = 8),
σ1s2, σ* 1s2, σ2s2s2, σ* 2s2
Hence, the bond order of Be2 is -(Nb – Na).
where,
Nb = Number of electrons in bonding orbitals.
Na – Number of electrons in anti-bonding orbitals.
∴ Bond order of Be2 = \(\frac{1}{2}\) (4 – 4) = 0
A negative or zero bond order means that the molecule is unstable. Hence, Be2 molecule does not exist.
Question 36.
Compare the relative stability of the following species and indicate their magnetic properties ; \(\mathbf{O}_{2}, \mathbf{O}_{2}^{+}, \mathbf{O}_{2}^{-}\)(superoxide), \(\mathrm{O}_{2}^{2-}\) (peroxide)
Answer:


![]()
Question 37.
Write the significance of a plus and a minus sign shown in representing the orbitals.
Answer:
Molecular orbitals are represented by wave function. A plus sign in an orbital indicates a positive wave function while a minus sign in an orbital represents a negative wave function. Combination of two wave functions having similar sign gave bonding molecular orbital while that having opposite sign gave antibonding molecular orbital.
Question 38.
Describe the hybridisation in case of PCl5. Why are the axial bonds longer as compared to equatorial bonds?
Answer:
The ground state and excited state outer electronic configurations of phosphorus (Z = 15) are as follows :
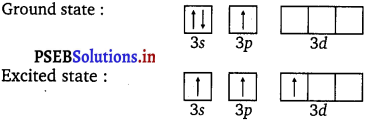
Phosphorus atom is sp3 d hybridised in the excited state. These orbitals are filled by the electron pairs donated by five Cl atoms as :
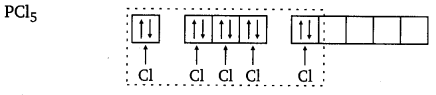
The five sp3d hybrid orbitals are directed towards the five corners of the trigonal bipyramidals. Hence, the geometry of PCl5 can be represented as :
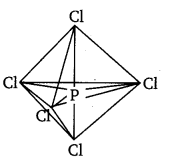
There are five P—Cl sigma bonds in PCl5. Three P—Cl bonds lie in one plane and make an angle of 120° with each other. These bonds are called equatorial bonds.
The remaining two P—Cl bonds lie above and below the equatorial plane and make an angle of 90° with the plane. These bonds are called axial bonds.
As the axial bond pairs suffer more repulsion from the equatorial bond pairs, axial bonds are slightly longer than equatorial bonds.
Question 39.
Define hydrogen bond. Is it weaker or stronger than the van der Waals forces?
Answer:
A hydrogen bond is defined as an attractive force between the hydrogen attached to an electronegative atom of one molecule and an electronegative atom of a different molecule (may be of the same kind). Due to a difference between electronegativities, the bond pair between hydrogen and the electronegative atom gets drifted far away from the hydrogen atom. As a result, a hydrogen atom becomes electropositive with respect to the other atom and acquires a positive charge.
![]()
The magnitude of H-bonding is maximum in the solid state and minimum in the gaseous state.
There are two types of H-bonds:
(i) Intermolecular H-bonds e.g., HF, H2O etc
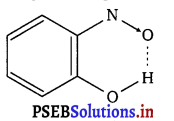
(ii) Intramolecular H-bonds e.g., o-nitrophenol
Hydrogen bonds are stronger than Van der Waals forces since hydrogen bonds are regarded as an extreme form of dipole-dipole interaction.
Question 40.
What is meant by the term bond order? Calculate the bond order of: \(\mathrm{N}_{2}, \mathrm{O}_{2}, \mathrm{O}_{2}^{+}\) and \(\mathbf{O}_{2}^{-}\).
Answer:
Bond order is defined as half of the difference between the number of electrons present in the bonding and anti-bonding orbitals of a molecule.
Bond order = \(\frac{1}{2}\) (Nb – Na)
If Nb > Na, then the molecule is said be stable. However, if Nb ≤ Na, then the molecule is considered to be unstable.
Bond order values 1, 2 or 3 correspond to single, double or triple bonds respectively.
Calculation of the bond order of \(\mathrm{N}_{2}, \mathrm{O}_{2}, \mathrm{O}_{2}^{+}\) and \(\mathbf{O}_{2}^{-}\).
Electronic configuration of N2
![]()

