Punjab State Board PSEB 12th Class Chemistry Important Questions Chapter 9 Coordination Compounds Important Questions and Answers.
PSEB 12th Class Chemistry Important Questions Chapter 9 Coordination Compounds
Very Short Answer Type Questions
Question 1.
Why is CO a stronger ligand than Cl– ?
Answer:
CO forms π bonds so it is a stronger ligand than Cl–.
Question 2.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
Answer:
When white light falls on the complex, some part of it is absorbed. Higher the crystal field splitting, lower will be the wavelength absorbed by the complex. The observed colour of complex is the colour generated from the wavelength left over.
Question 3.
How many isomers are there for octahedral complex [CoCl2 (en) (NH3)2]+?
Answer:
There will be three isomers: cis and trans isomers. Cis will also show optical isomerism.
![]()
Question 4.
Why are low spin tetrahedral complexes not formed?
Answer:
Because for tetrahedral complexes, the crystal field stabilisation energy is lower than pairing energy.
Question 5.
A complex of the type [M(AA)2X2]n+ is known to be optically active. What does this indicate about the structure of the complex? Give one example of such complex.
Answer:
An optically active complex of the type [M(AA)2X2]n+ indicates cis- octahedral structure, e.g., cis-[Pt(en)2Cl2]2+ or cis-[Cr(en)2Cl2]+.
Question 6.
Why is the complex [Co(en)3]3+ more stable than the complex [CoF6]3-?
Answer:
Due to chelate effect as the complex [Co(en)3]3+ contains chelating ligand \(\ddot{\mathrm{NH}}_{2}-\mathrm{CH}_{2}-\mathrm{CH}_{2}-\ddot{\mathrm{NH}}_{2}\).
![]()
Question 7.
What do you understand by ‘denticity of a ligand’?
Answer:
The number of coordinating groups present in ligand is called the denticity of ligand. For example, denticity of ethane-1, 2-diamine is 2, as it has two donor nitrogen atoms which can link to central metal atom.
Question 8.
What type of isomerism is shown by the complex [CO(NH3)5(SCN)]2+?
Answer:
Linkage isomerism.
Question 9.
Arrange the following complex ions in increasing order of crystal field splitting energy △0 :
[Cr(Cl)6]3-, [Cr(CN)6]3-, [Cr(NH3)6]3+
Answer:
[Cr(Cl)6]3- < [Cr(NH3)6]3+ < [Cr(CN)6]3-
![]()
Question 10.
A coordination compound with molecular formula CrCl3.4H2O precipitates one mole of AgCl with AgNO3 solution. Its molar conductivity is found to be equivalent to two ions. What is the structural formula and name of the compound?
Answer:
[Cr(H2O)4Cl2] Cl
[Tetraaquadichloridochromium (III) chloride]
Short Answer Type Questions
Question 1.
Give the electronic configuration of the following complexes on the basis of crystal field splitting theory.
[CoF6]3-, [Fe(CN)6]4- and [Cu(NH3)6]2+
Answer:
[CoF6]3-: Co3+(d6) \(t_{2 g}^{4} e_{g}^{2}\)
[Fe(CN)6]4- : Fe2+ (d6) \(t_{2 g}^{6} e_{g}^{0}\)
[Cu(NH3)6]2+ : Cu2+ (d9) \(t_{2 g}^{6} e_{g}^{3}\)
![]()
Question 2.
(i) What type of isomerism is shown by [Co(NH3) 5ONO]Cl2?
(ii) On the basis of crystal field theory, write the electronic configuration for d4 ion, if △0 < P.
(iii) Write the hybridisation and shape of [Fe(CN)6]3-.
(Atomic number of Fe = 26)
Answer:
(i) Linkage isomerism and the linkage isomer is [Co(NH3) 5ONO]Cl2.
(ii) If △0 < P, the fourth electron enters one of two eg orbitals giving the configuration \(t_{2 g}^{3} e_{g}^{1}\).
(iii) Fe3+ : 3d5 4s0
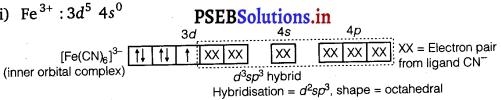
Question 3.
Explain why [Fe(H2O)6]3+ 5.92 BM whereas [Fe(CN)6]3- has a value of only 1.74 BM.
Answer:
[Fe(CN)6]3- involves d2sp3 hybridisation with one unpaired electron and [Fe(H2O)6]3+ involves sp3d2 hybridisation with five unpaired electrons. This difference is due to the presence of strong CN– and weak ligand H2O in these complexes.
![]()
Question 4.
CuSO4∙5H2O is blue in colour while CuSO4 is colourless. Why?
Answer:
In CuSO4∙5H2O, water acts as ligand as a result it causes crystal field splitting. Hence, d-d transition is possible in CuSO4∙5H2O and shows colour. In the anhydrous CuSO4 due to the absence of water (ligand), crystal field splitting is not possible and hence it is colourless.
Question 5.
Why do compounds having similar geometry have different magnetic moment?
Answer:
It is due to the presence of weak and strong ligands in complexes, if CFSE is high, the complex will show low value of magnetic moment and vice versa, e.g., [CoF6]3- and [Co(NH3)6]3+, the former is paramagnetic and the latter is diamagnetic.
Question 6.
A metal ion Mn+ having d4 valence electronic configuration combines with three bidentate ligands to form a complex compound. Assuming △0 > P:
(i) Write the electronic configuration of d4 ion.
(ii) What type of hybridisation will Mn+ ion has?
(iii) Name the type of isomerism exhibited by this complex.
Answer:
(i) \(t_{2 g}^{4} e_{g}^{0}\)
(ii) sp3d2
(iii) Optical isomerism
![]()
Long Answer Type Questions
Question 1.
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following: [COF6]3-, [CO(H2O)6]2+, [CO(CN)6]3
Answer:
Magnetic moment, μ = \(\sqrt{n(n+2)}\)
Where, n = Number of unpaired electrons
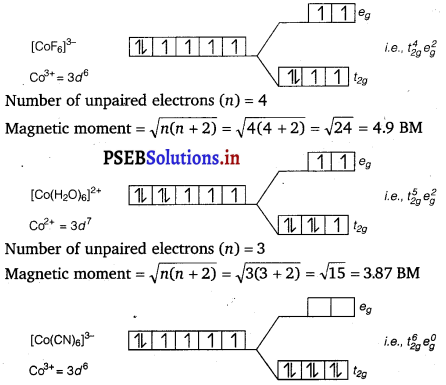
No unpaired electrons, so it is diamagnetic.
![]()
Question 2.
(i) Draw the geometrical isomers of complex [Pt(NH3)2Cl2].
(ii) Write the hybridisation and magnetic behaviour of the complex [Ni(CO)4].
(Atomic no. of Ni = 28)
Answer:
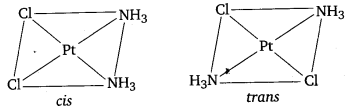
Geometrical isomers of [Pt(NH3)2Cl2]
(ii) The complex [Ni(CO)4] involves sp3 hybridisation.
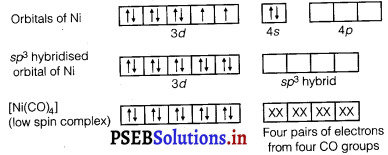
The complex is diamagnetic as evident from the absence of unpaired electrons.
