Punjab State Board PSEB 12th Class Chemistry Important Questions Chapter 13 Amines Important Questions and Answers.
PSEB 12th Class Physics Important Questions Chapter 13 Amines
Very short answer type questions
Question 1.
CH3NH2 is more basic than C6H5NH2 why?
Answer:
Aliphatic amines (CH3NH2) are stronger bases than aromatic amines (C6H5NH2) because due to resonance in aromatic amines, the lone pair of electrons on the nitrogen atom gets delocalized over the benzene ring and thus is less easily available for protonation.
Question 2.
What is the role of pyridine in the acylation reaction of amines? [NCERT Exemplar]
Answer:
Pyridine and other bases are used to remove the side product, L e., HCl from the reaction mixture.
Question 3.
A primary amine, RNH2 can be reacted with CH3 – X to get secondary amine, R-NHCH3 but the only disadvantage is that 3° amine and quaternary ammonium salts are also obtained as side products. Can you suggest a method where RNH2 forms only 2° mine? [NCERT Exemplar]
Answer:
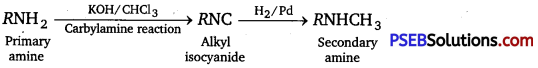
Primary amines show carbylamine reaction in which two H-atoms attached to N-atoms of NH2 are replaced by one C-atom. On catalytic reduction, isocyanide (formed) produces secondary amine and not tertiary or quaternary salts.
Question 4.
What is Hinsberg reagent? (NCERT Exemplar)
Answer:
Benzene sulphonyl chloride (C6H5SO2Cl) is known as Hinsberg reagent. It is used to distinguish between primary, secondary and tertiary amine.
![]()
Question 5.
The conversion of primary aromatic amines into diazonium salts is known as……
Answer:
Diazotisation.
Question 6.
Rearrange the following in an increasing order of their basic strengths:
C6H5NH2,C6H5N(CH3)2,(C6H5)2NH and CH3NH2
Answer:
(C6H5)2NH < C6H5NH2 <C6H5N(CH3)2 <CH3NH2
Question 7.
What is the best reagent to convert nitrile to primary amine? [NCERT Exemplar]
Answer:
Reduction of nitriles with sodium/alcohol or LiAlH4 gives primary amine.
Question 8.
Suggest a route by which the following conversion can be accomplished: [NCERT Exemplar]
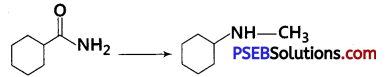
Answer:
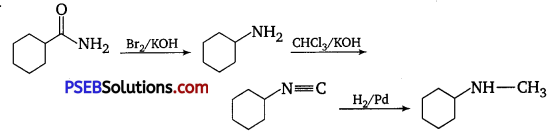
Question 9.
What is the role of HNO3 in the nitrating mixture used for the nitration of benzene? [NCERT Exemplar]
Answer:
HNO3 acts as a base in the nitrating mixture and provides the electrophile NO2.
Question 10.
Why is benzene diazonium chloride not stored and is used immediately after its preparation? [NCERT Exemplar]
Answer:
Benzene diazonium chloride is very unstable.
Short answer type questions
Question 1.
Write the structures of A, B and C in the following:
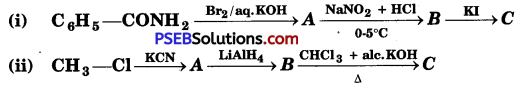
Answer:
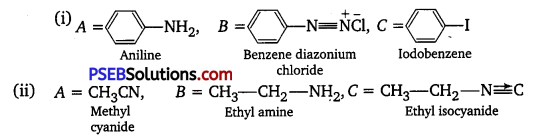
Question 2.
Give a chemical test to distinguish between C6H5CH2NH2 and C6H5NH2.
Answer:
C6H5CH2NH2 reacts with HNO2 at 273-278 K to give diazonium salt, which being unstable decomposes with brisk evolution of N2 gas.
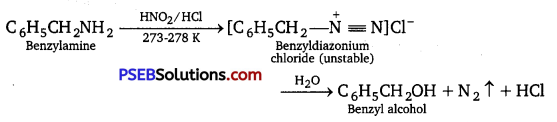
whereas, C6H5NH2 reacts with HNO2 at 273-278 K to form stable benzene diazonium chloride, which upon treatment with an alkaline solution of f3-naphthol, gives an orange dye.
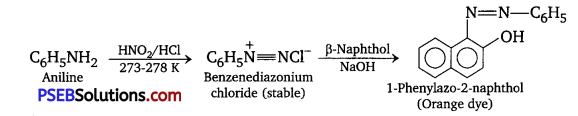
![]()
Question 3.
Complete the following reaction:  [NCERT Exemplar]
[NCERT Exemplar]
Answer:
The reaction exhibits an azo-coupling reaction of phenols. Benzene diazonium chloride reacts with phenol in such a manner that the para position of phenol is coupled with diazonium salt to form p-hydroxy azobenzene.
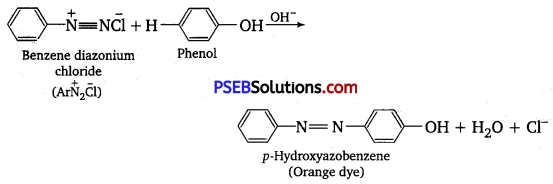
Question 4.
A solution contains 1 g mol. each of p-toluene diazonium chloride and p-nitrophenyl diazonium chloride. To this lg mol. of alkaline solution of phenol is added. Predict the major product. Explain your answer. [NCERT Exemplar]
Answer:
The above-stated reaction is an example of electrophilic aromatic substitution. In alkaline medium, phenol generates phenoxide ion which is more electron-rich than phenol and more reactive for electrophilic attack. The electrophile in this reaction is aryldiazonium cation. As we know, stronger the electrophile faster is the reaction. p Nitrophenyldiazonium cation is a stronger electrophile than p-toluene diazonium cation. So, nitrophenyl diazonium chloride couples preferentially with phenol
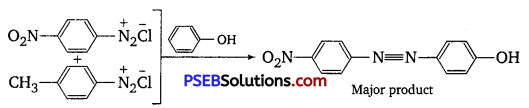
Question 5.
Under what reaction conditions (acidic, basic) the coupling reaction of aryl diazonium chloride with aniline is carried out? [NCERT Exemplar]
Answer:
In strongly basic conditions, benzene diazonium chloride is converted, into diazohydroxide and diazoate as both of which are not electrophilic and do not couple with aniline.

Similarly, in highly acidic conditions, aniline gets converted into an anilinium ion. From this, result aniline is no longer nucleophilic acid and hence will not couple with diazonium chloride. Hence, the reaction is carried out under mild conditions, i.e., pH 4-5
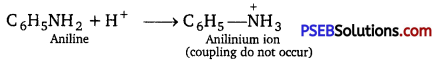
Question 6.
An organic aromatic compound ‘A’ with the molecular formula C6H7N is sparingly soluble in water. ‘A’ on treatment with dil. HCl gives a water-soluble compound ‘B’ ‘A’ also reacts with chloroform in presence of alcoholic KOH to form an obnoxious smelling compound ‘C’. ‘A’ reacts with benzene sulphonyl chloride to form an alkali-soluble compound ‘D’ ‘A’ reacts with NaNO2 and HCl to form a compound ‘E’ which on reaction with phenol forms an orange-red dye ‘F’ Elucidate the structures of the organic compounds from ‘A’ to ‘F’
Answer:
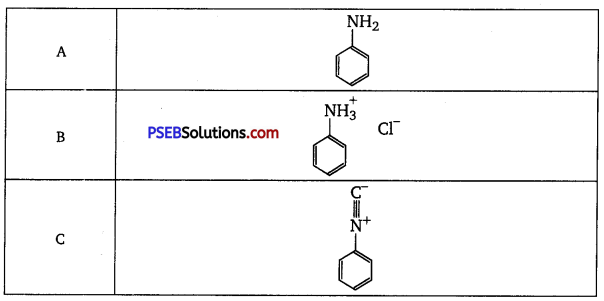
Long answer type questions
Question 1.
Predict the reagent or the product in the following reaction sequence : [NCERT Exemplar]
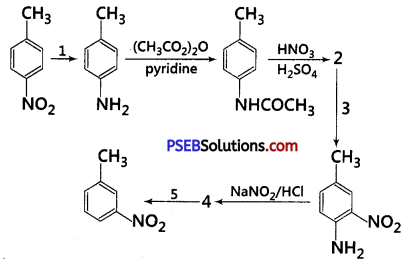
Answer:
A correct sequence can be represented as follows including all reagents:
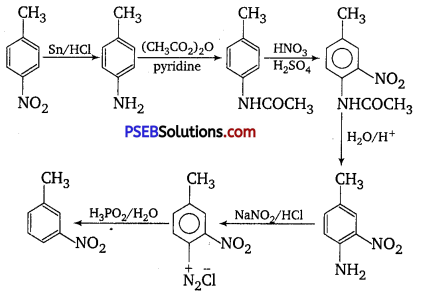
Hence,
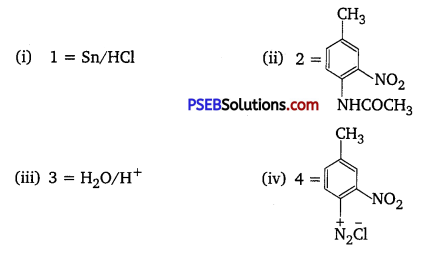
(v) 5 = H3PO2/H2O
![]()
Question 2.
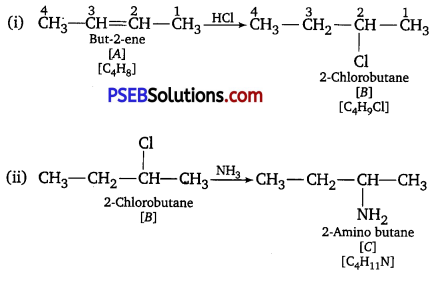
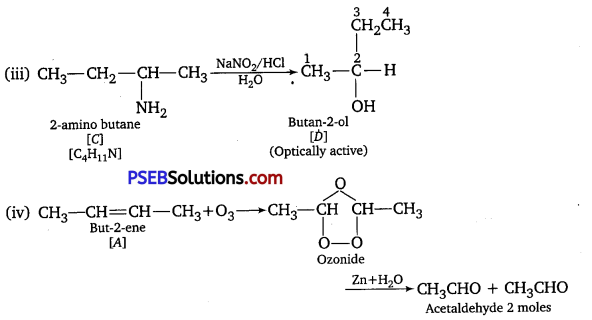
A hydrocarbon ‘A’ (C4H8) on reaction with HC1 gives a compound ‘B’, (C4H9Cl), which on reaction with 1 mol of NH3 gives compound ‘C’, (C4H11N). On reacting with NaNO2 and HCl followed by treatment with water, compound ‘C’ yields an optically active alcohol, ‘D’ Ozonolysis of ‘A’ gives 2 mols of acetaldehyde. Identify compounds ‘A’ to ‘D\ Explain the reactions involved. [NCERT Exemplar]
Answer:
(i)Addition of HCl to compound A shows that compound A is alkene.
Compound ‘B’ is C4H9Cl.
(ii) Compound‘B’reacts with NH2. It forms amine‘C’.
![]()
(iii) ‘C’ gives diazonium salt with NaNO2/HCl, which yields an optically active alcohol.
So, ‘C’ is aliphatic amine.
(iv) ‘A on ozonolysis produces 2 moles of CH3CHO. So, A is CH3– CH =CH-CH3 (But-2-ene).
Reactions